 
As was reported in the 2020 CoSTR, all observational studies previously identified were at critical risk of bias due to confounding, and individual results are thus difficult to interpret. No new observational studies were identified since the prior review. This updated systematic review identified one additional RCT (Cheskes 2022 1). This CoSTR is an update of the 2020 systematic review (Deakin 2020 ). PROSPERO Registration Submitted OctoConsensus on Science Literature search for this update included studies published from Februto November 7, 2022. Timeframe: All years and all languages were included as long as there was an English abstract unpublished studies (e.g., conference abstracts, trial protocols) were excluded. All relevant publications in any language are included as long as there is an English abstract. 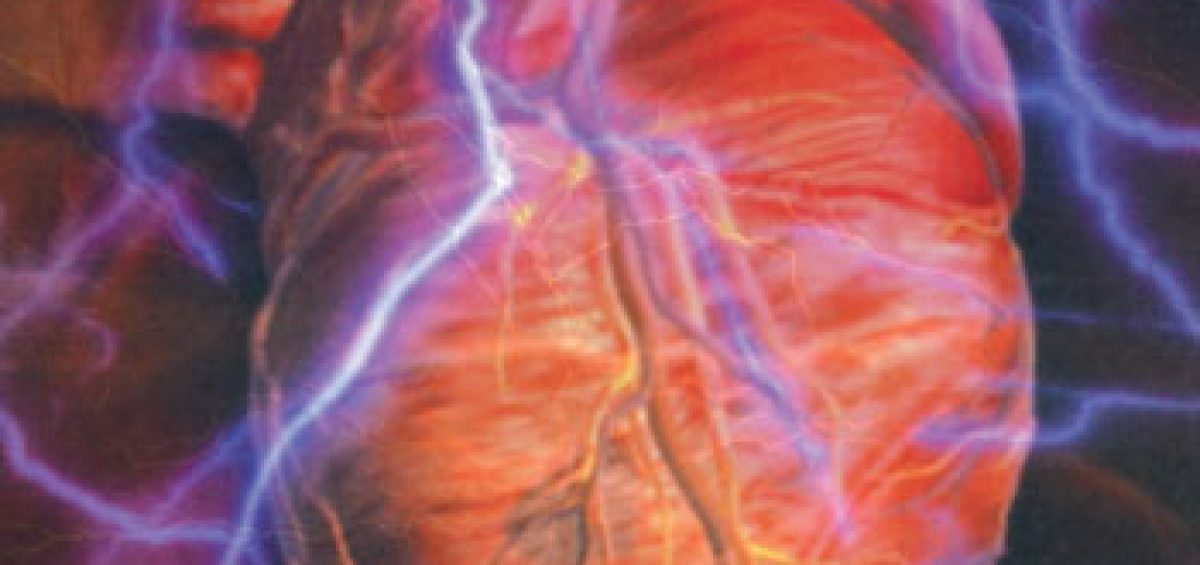
Unpublished studies (e.g., conference abstracts, trial protocols) are excluded. Study Designs: Randomized controlled trials (RCTs) and non-randomized studies (non-randomized controlled trials, interrupted time series, controlled before-and-after studies, cohort studies) are eligible for inclusion. 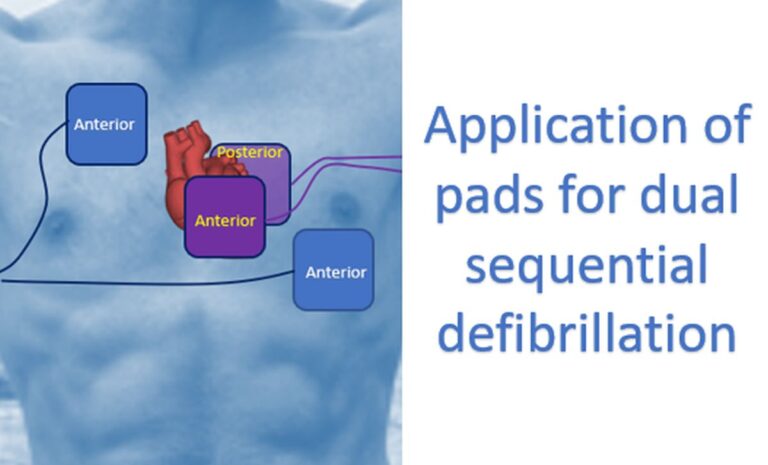
Important – Return of Spontaneous Circulation (ROSC), survival to hospital admission. Outcomes: Critical – Survival to hospital discharge or good neurological survival at discharge/30 days, or greater than 30 days. Intervention: Double sequential defibrillation strategyĬomparators: Standard defibrillation strategy Population: Adults in any setting (in-hospital or out-of-hospital) with cardiac arrest and a shockable VF/pVT cardiac arrest rhythm. The PICOST (Population, Intervention, Comparator, Outcome, Study Designs and Timeframe) 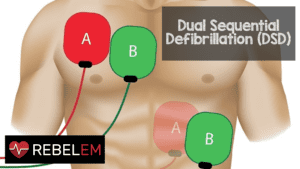
(PROSPERO registration CRD42022365305, registered on October 17, 2022) These data were taken into account by the task force when formulating the treatment recommendations. Due to the publication of a new RCT, an updated systematic review was performed. The continuous evidence evaluation process for the production of Consensus on Science with Treatment Recommendations (CoSTR) started with a systematic review conducted on behalf of the ALS task force in 2020. Double Sequence Defibrillation, Consensus on Science with Treatment Recommendations Brussels, Belgium: International Liaison Committee on Resuscitation (ILCOR) Advanced Life Support Task Force, 2022 December 3. Ohshimo S, Drennan I, Deakin CD, Soar J, Berg KM on behalf of the International Liaison Committee on Resuscitation Advanced Life Support Task Force. He declared his intellectual COI during all discussions about the included trial and draft treatment recommendations. The following Task Force members and other authors declared an intellectual conflict of interest and this was acknowledged and managed by the Task Force Chairs and Conflict of Interest committees: Ian Drennan was a co-investigator on the RCT of defibrillation strategies (Cheskes 2022) included in this systematic review, and therefore did not participate in the study selection or bias assessment steps of the review. The following Task Force members and other authors were recused from the discussion as they declared a conflict of interest: none The ILCOR Continuous Evidence Evaluation process is guided by a rigorous ILCOR Conflict of Interest policy. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
